Lactose properties and behavior in 3D powder bed printing

Working with the Netherlands Organization for Applied Scientific Research (TNO), DFE Pharma tested 20 lactose grades in 3D powder bed printing. The primary objective of this study was to select lactose with sufficient flow, wetting and binding to be used effectively in the powder bed 3D printing of pharmaceuticals.
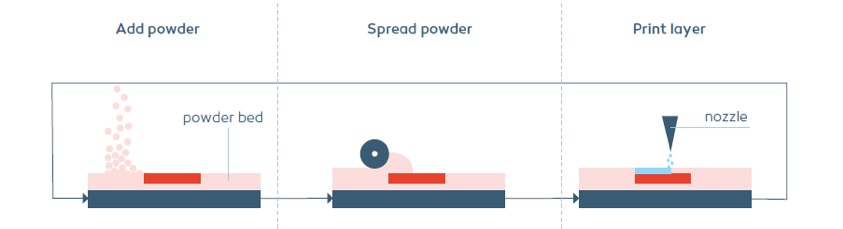
Powder bed printing enables to formulate sensitive API’s without exposing them to heat or excessive moisture. It also creates IR formulations with fast and complete API release. Additionally, the only FDA approved 3D printed drug Spritam is produced by powder bed printing.

DFE pharma is sharing its knowledge and results on the study in the whitepaper.
Please check your email after launching the request as it might be held in your SPAM

