Lipid Based Excipients for Modified Release
Modified- or sustained-release matrices are designed to release a drug slowly over an extended period of time. Formulations must be robust to ensure drug release over time and avoid dose-dumping. Lipid-based matrices are water-insoluble and do not swell or erode when in contact with aqueous media. When used as sustained-release agents, they form an inert matrix from which the drug diffuses slowly.
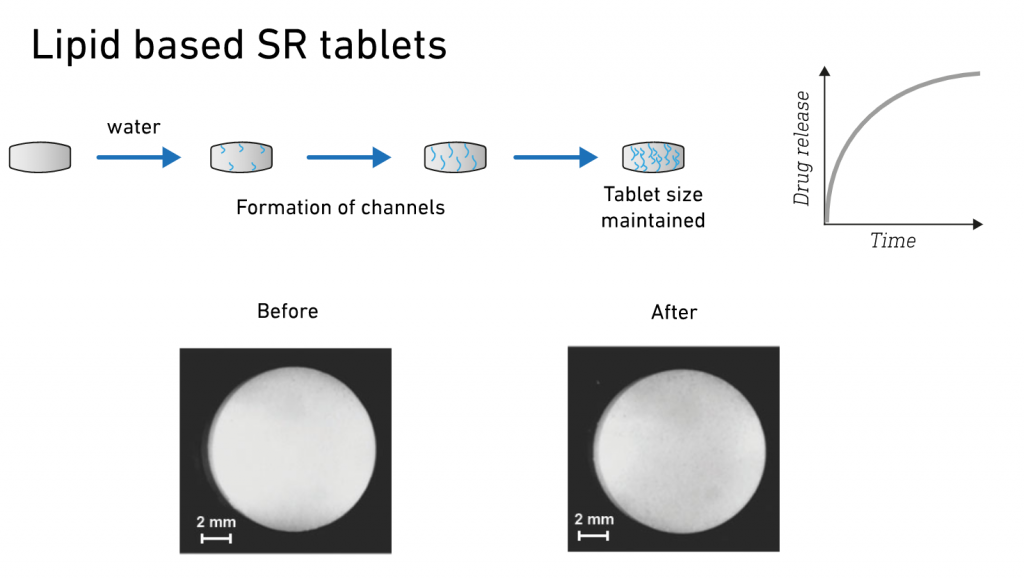
Advantages of Gattefossé’s modified-release agents:
• Drug release is a function of diffusion, which enables simple product design and offers both straightforward modulation of drug release and high reproducibility.
• Tablets are biopharmaceutically robust as the lipid matrix is not sensitive to physiological variations (e.g., pH, digestive processes, alcohol level).
• Agents are suitable for a wide range of processing techniques, including direct compression, granulation, melt coating, hot melt extrusion, and capsule molding.
The information on the topic of Lipid-based drug delivery is taken from the chapter
“Excipients for Modified Release” from the Gattefossé website.
Range of modified release agents
Compritol® 888 ATO
- finely atomized lipid excipient powder of well-defined, reproducible composition and particle size
- used at proportion of 10%–40%, depending on drug solubility and diluent properties
- suitable for direct compression, granulation, and hot processes (i.e., hot melt coating, granulation, and extrusion)
- crystalline material with narrow melting range (∼70 °C) and rapid recrystallization, ideal for hot melt processes
- GRAS status and precedence of use with many APIs (FDA IID)
Precirol® ATO 5
- finely atomized lipid excipient powder of well-defined, reproducible composition and particle size
- crystalline material with narrow melting range (∼55 °C) and rapid recrystallization, suitable for use in hot processes (i.e., hot melt coating, extrusion, and capsule molding)
- GRAS status and precedence of use with many APIs (FDA IID)
Geleol™ Mono and Diglycerides NF
- available in pellet form
- crystalline material with narrow melting range (∼60 °C) and rapid recrystallization, suitable for use in hot processes (i.e., hot melt coating, extrusion, and capsule molding)
- GRAS status and precedence of use with many APIs (FDA IID)
Source: Gattefosse Website chapter “Excipients for Modified Release”

