Development, recent advances, and updates in binary, ternary co-amorphous systems, and ternary solid dispersions
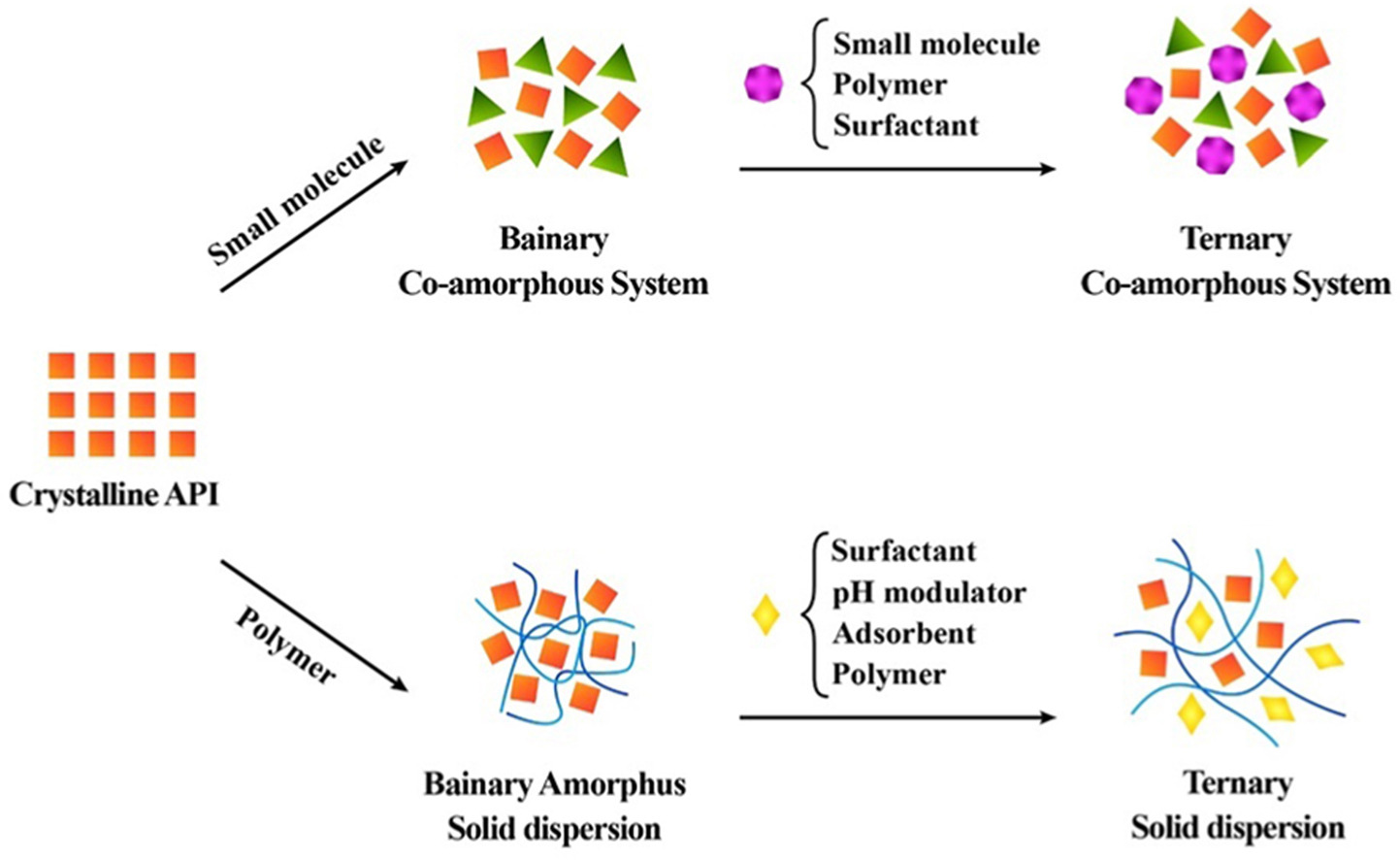
Binary co-amorphous systems (CAMS) are a type of solid dispersion containing an active pharmaceutical ingredient (API) and small molecule excipients. The properties of these substances make them novel and a very demanding aspect of pharmaceutical research because they improve solubility, physical stability, and bioavailability in poorly soluble drugs in comparison to their pure crystal or amorphous counterparts. Binary CAMS may however not satisfy all criteria related to dissolution, solubility, recrystallization prevention, and stability. As a result, ternary systems incorporating a third ingredient in CAMS offer several advantages in terms of stability and drug solubility.
Similarly, ternary systems in the context of amorphous solid dispersion (ASD) improve the drug’s stability and solubility over binary solid dispersions. During this review, we summarize recent findings regarding ternary CAMS, ternary solid dispersion (TSD) and binary CAMS. Recent advances in the preparation, mechanism of stabilization, and in-vitro and in-vivo performances of binary and ternary systems are also discussed along with their final dosage forms.
Table 3
Recent studies on ternary solid dispersion (TSD) since 2016. TPGS: tocopheryl polyethylene glycol 1000 succinate, SDS: Sodium dodecyl sulfate, SLS: Sodium lauryl sulfate, HPMCP: hydroxypropyl methylcellulose phthalate, HPMC: hydroxypropyl methylcellulose, PEG: polyethylene glycol, HP-b-CD: 2-Hydroxypropyl-beta-cyclodextrin, PVP: poly vinyl pyrrolidone, HPMCAS: hypromellose acetate succinate, HME: hot melt extrusion.
| Binary solid dispersion | Third ingredient | The additional ingredient’s type | Outcomes | Method |
|---|---|---|---|---|
| LW6 (3-[2-(4-adamantan-1-yl-phenoxy)- acetylamino]-4-hydroxy-benzoic acid methyl ester)-Povidone K30 | Poloxamer 407 | Polymer | Increase solubility | Solvent evaporation |
| Lurasidone - Poloxamer 188 | Florite | Adsorbent | Inhibited transition from an amorphous to the crystalline form and enhanced stability of amorphous form. | Fusion method |
| Carbamazepine-Kollidon® VA64 | Neusilin UFL2 | Adsorbent | Enhanced the dissolution and carbamazepine flowability. | Rotary evaporation |
| Celecoxib -Phosphatidylcholine (PC) | Neusilin US2 | Adsorbent | Improved flowability | Solvent evaporation |
| Itraconazole- HPMCP | Soluplus | Polymer | Increase solubility, stability and processability, | Hot melt extrusion/Spray drying |
| Nateglinide-Poloxamer 188 | Na2CO3 | pH modulator | Boosting nateglinide’s bioavailability by up to four times | melt dispersion |
| Abiraterone- HP-b-CD | HPMCAS | Polymer | Increase Precipitation inhibition | KinetiSol® technology |
| Tectorigenin- PVP | PEG4000 | Polymer | Increase solubility | Rotary evaporation |
| Dipyridamole -Eudragit® S100 | Tartaric acid | pH modulator | Rapid dissolution of dipyridamole | Freeze-drying |
| Glycyrrhetinic acid- PVP | l-arginine/ meglumine | pH modulator | enhance ionization, thereby increasing solubility | HME |
| Cinnarizine- Soluplus | HPMC/PVP | Polymer | Increase Solubility and stability | HME |
| Nifedipine- HPMC | Eudragit®-S | Polymer | Increase Solubility | Spray drying |
| Bedaquiline fumarate-Poloxamer 188 | TPGS | Surfactant | Increase solubility, dissolution rate and permeability | Solvent evaporation |
| Ritonavir- HPMCAS | SLS | Surfactant | Inhibit recrystallization | Solvent evaporation |
| Loratadine-shellac | HPMC | Polymer | Increase solubility and supersaturation of loratadine | HME |
| Chlorthalidone -Soluplus | SLS | Surfactant | 5.5 times more solubility compared with the pure drug | Spray dried |
| Felodipine -Soluplus | SDS | Surfactant | Increased dissolution | HME |
| Ezetimibe- PVP K30 | Poloxamer 188 | Surfactant | Improved solubility and supersaturation in the bio-relevant media. | Melt quenching |
Download the full article as PDF here Development, recent advances, and updates in binary, ternary co-amorphous systems, and ternary solid dispersions
or read it here
Arezoo Saberi, Maryam Kouhjani, Davood Yari, Afsaneh Jahani, Kofi Asare-Addo, Hossein Kamali, Ali Nokhodchi,
Development, recent advances, and updates in binary, ternary co-amorphous systems, and ternary solid dispersions,
Journal of Drug Delivery Science and Technology, Volume 86, 2023, 104746, ISSN 1773-2247, https://doi.org/10.1016/j.jddst.2023.104746.
Watch the video and read more on “Vitamin E TPGS by PMC Isochem” here:

