Investigating the benefits of delivering gastric resistant functionality to a tablet by applying a colourless top-coat based on Kollicoat® MAE 30 DP
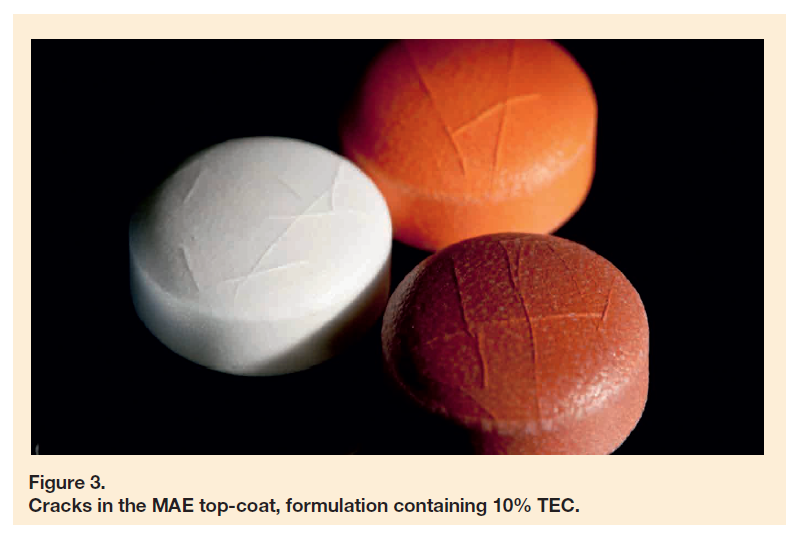
In the field of gastric resistant film-coating, a lot of obstacles can occur during formulation development. Some pharmaceutical actives (e.g. omeprazole) are prone to interactions with the functional polymer poly(methacrylic acid-co-ethyl acrylate) (MAE). Yet, one should consider that the coating material itself is also vulnerable to interactions with excipients (such as pigments or colourants) present in the coating formulation.
This work was set to investigate whether the gastric resistant functionality can be applied with a colourless film, coated onto a tablet bearing a coloured instant release sub-coat.
Click image to enlarge: MORE ON BASF
MORE ON BASF
See our webinar and have the first live explanation of the new Enteric-Release Coatings Module in ZoomLab™


