Suppression of Related Substance in Tablets Containing Pressure-Sensitive Drugs with Elongated Microcrystalline Cellulose

This poster was presented at the PBP World Meeting in Vienna 2024 by Asahi-Kasei
INTRODUCTION
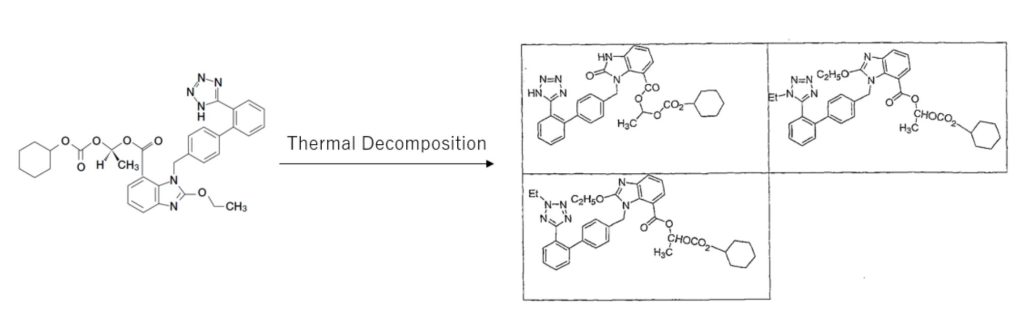
- It is known that the pressure, friction, heat, etc. applied during compression molding of some drugs causes crystal distortion and destabilization.
- When dealing with such kinds of substances, excipient selection is important.
- In this study, we report the use of different types of MCCs (Ceolus PH, Ceolus KG) to investigate their applicability in tablets containing candesartan cilexetil (CC) as a model pressure-sensitive drug.
Ceolus Microcrystalline cellulose (MCC)
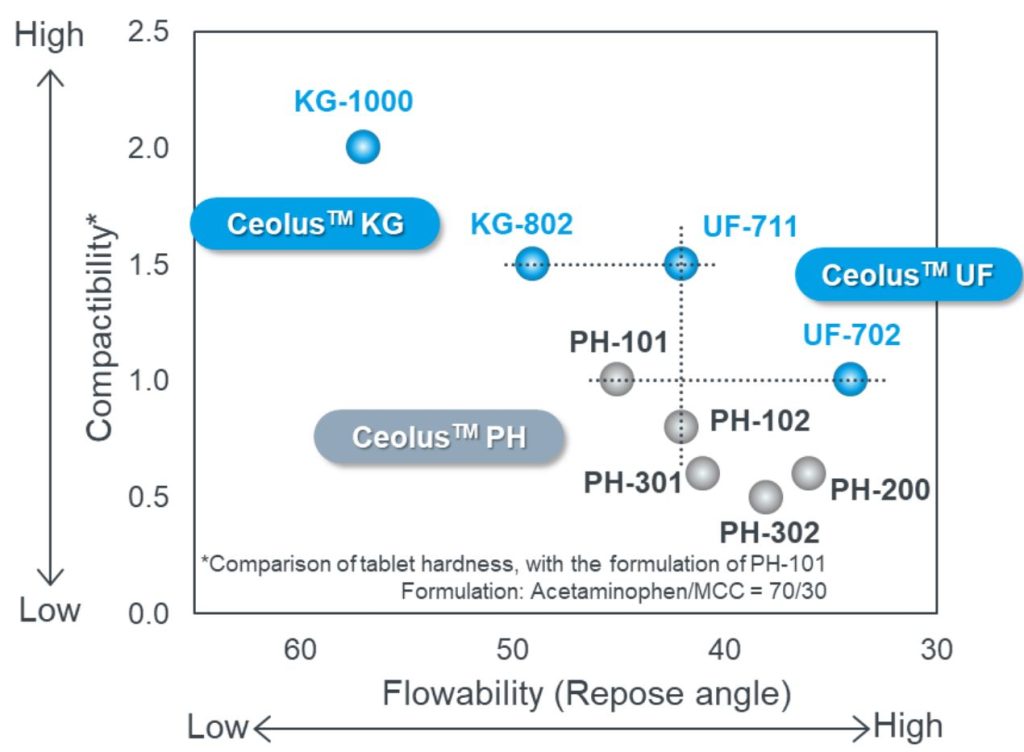
Ceolus brings unique properties due to their particle shapes.
Ceolus KG
Ceolus KG is a highly compactible MCC with elongated particles.
- Solves tableting issues
- Insufficient hardness, sticking, capping, high friability

EXPERIMENTS
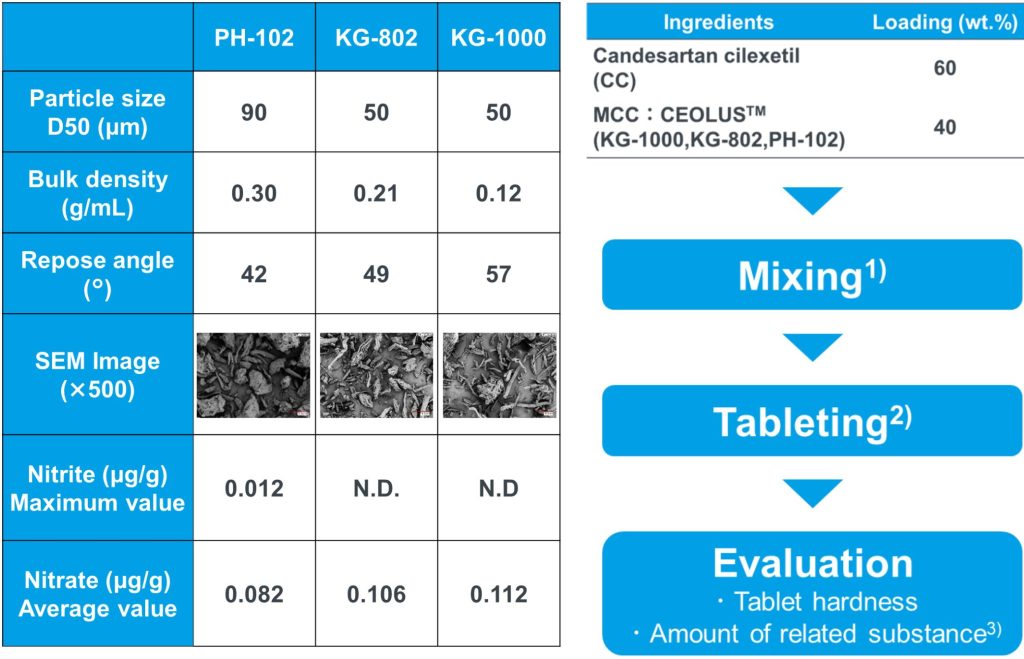
Table 1. Properties of MCCs used in this study.
1) Mixing condition: PE bag, Mixing time: 3 min.
2) Tool: MODEL-1011 CREEP, AIKOH ENGINEERING
Tablet size: 200mg, 8mmΦ-30R
*Tableting was carried out under the static condition
(10 sec of creep holding time).
*Compression force was adjusted to obtain the tablet
hardness of 50 N that is practically required value.
3) Tool: Model LC-10ADVP, Shimadzu Corporation
*The tablets were dissolved in a mobile phase to a
drug concentration of 160 mg/L, then filtered through a
0.45 μm filter.
*The ratio of related compounds was quantified by the
peak area ratio of CC and its related-compounds.
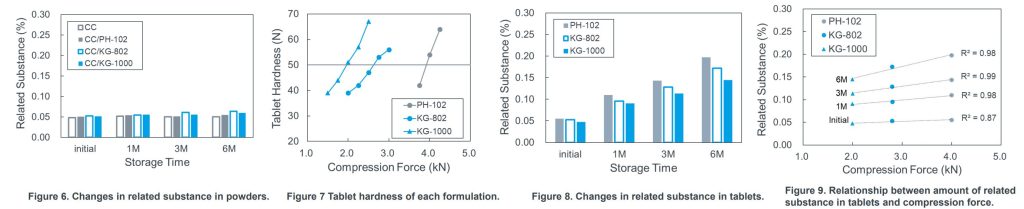
RESULTS
CONCLUSION
- The tableting pressure to obtain tablets of the same hardness could be decreased with applying elongated-shaped MCCs (KG-1000 and KG-802) compared to amorphous-shaped MCC (PH-102).
- The lower the compression force was applied, the less the amount of related substance generated.
- KG-1000 showed the least amount of related substance.
See the full poster on “Suppression of Related Substance” here
(click the picture to download the poster)
Source: Lei, J., Zhang, X., Zhuo, Z., Zhu, K., Sun, P., and Fan, Q., Asahi Kasei, HPLC-UV simultaneous determination of candesartan cilexetil and hydrochlorothiazide in compound candesartan cilexetil tablets, Yaow u Fenxi Zazhi, 27 (4), 566-568 (2007), Japanese Patent Publication 2008-505935A, poster “Suppression of Related Substance”, PBP 14th World Meeting, 18 – 21 March 2024, Vienna
Do you want to see more posters of the PBP World Meeting 2024 in Vienna?